Automotive Sustainability: Desulfurization and Catalytic Converters
Written by: Jack Morby
There are millions of cars in use every day that contribute 74.5 percent of the 8 billion tons of CO2 emissions that come from the transport sector (International Energy Agency, 2018). In return for comfortable and personalized travel, the environment suffers. Regardless, transportation by car is necessary for most people to make a living and get from one place to another. Taking the train or bike does not work for every case. Therefore, the most reasonable way to lower carbon dioxide emissions is developing new automotive innovations to make cars more sustainable. Fortunately, there are already engineering and design structures in place to facilitate these innovations. Since 1950, the levels of pollutants coming out of tailpipes have decreased by 98-99 percent according to the Environmental Protection Agency (EPA).The automotive industry is expansive and a great place to begin with developing higher quality sustainability practices.
Desulfurization
Over ninety percent of all cars on the road today run on gasoline or diesel fuel. Carbon monoxide, nitrous oxides, and carbon dioxide are the pollutants that are a problem for the environment. The quantity of pollutants in part relies on the quality of the fuel that the vehicle uses. For example, it is impossible to reduce air pollution without having sulfur-free fuel. Sulfur is a pollutant directly, but more importantly, sulfur prevents the adoption of all major pollution control technologies. The process of adsorption, the adhesion of a particle to a material’s surface, occurs between sulfur and platinum, palladium, and other rare metals. No significant air pollution reduction strategy can work without reducing sulfur to near-zero levels. Luckily in the United States, the EPA has installed legislation that requires gas to be sold with less than 10 ppm of sulfur, which is an incredible improvement—only eighteen years ago, this number had been 120 ppm.
More recent developments in 2017 include a research group at CalTech finding a new method to reduce the amount of sulfur in fuel even lower, to about 2 ppm. Named the “KOSi method”, this technique uses potassium salt, a cheap and abundant material. To break both bonds connecting the sulfur atom to the molecule, a trimethylsilyl functional group interacts with the molecule and “cleaves” the bond. The reaction also can be completed in low enough temperatures to be considered mild conditions, something necessary for the method to be viable on a broad scale. The figure below shows the molecular structure where the process can eliminate sulfur atoms. The next task for the team is to make the reaction ready for large-scale fuel production and implementation. Forcing the fuel sulfur levels to be as low as possible increases the lifespan of pollution control technologies, which is integral for environmental sustainability.
Figure 1
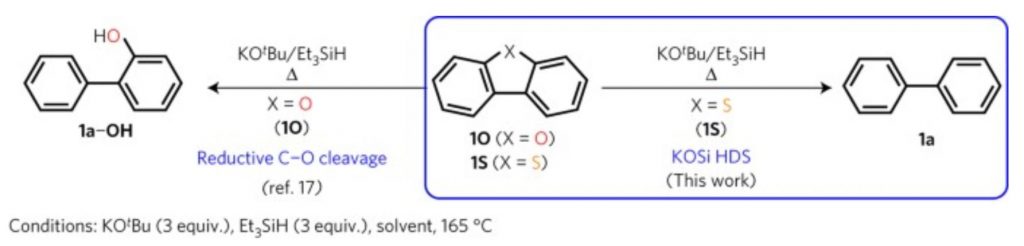
Reductive cleavage of C–X bonds by the KOSi system.
Source: Toutov, A. A., Salata, M., etc.
Catalytic Converter
The catalytic converter has garnered recent attention because of how it is often stolen out of the bottom of cars to make money off of platinum, rhodium, and palladium, the metals within. These precious metals quickly lose effectiveness if sulfur is present. The catalytic converter was a major invention from the nineteen-fifties by French engineer Eugene Houdry and was later made production-ready in 1973. The catalytic converter, which attaches onto the exhaust pipe, serves to remove pollutants such as carbon monoxide and nitrous oxides in addition to leftover fuel that did not combust. It helps eliminate all of the major pollutants, except for carbon dioxide. The process within the converter begins with platinum and rhodium completing a reduction reaction to pull the oxygen off of the nitrous oxides. After it pulls off the oxygen in the reduction reaction, it completes an oxidation reaction with platinum and palladium that attaches an oxygen atom to the carbon monoxide molecules. This creates carbon dioxide, a greenhouse gas that is three hundred times less toxic and harmful to the ozone layer than nitrous oxides. Finally, it causes any hydrocarbons (unburnt fuel) to be oxidized as well, making water and more carbon dioxide. The ceramic structure coated with precious metals is organized in a honeycomb pattern, as pictured below, which optimizes the surface area the metals have to interact with the gas without restricting airflow. The catalytic converter gets extremely hot during use and this is necessary for the reaction to reach activation energy and occur. Multiple studies over the years have even shown that in most cases the working catalytic converter does not affect fuel efficiency or horsepower significantly. (Gambarotta, 2019) This is promising because there are no negative effects to this invention’s use.
Figure 2
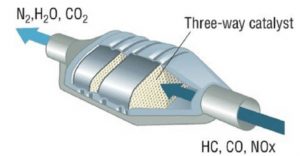
A three-way catalytic converter with the typical honeycomb structure and the gaseous inputs/outputs listed.
Source: Laurenzi, M. & Spigler, R.
Conclusion
The emission of greenhouse gasses and other pollutants from the exhaust of automobiles is detrimental to the health of the environment, but the innovations of the past and future are working to solve this problem. Whether it is catalytic converters, heralded as one of the most important environmental technology breakthroughs ever, or the coming age of electric and even hydrogen cars, they all play some part in the fight against pollution. Automotive companies have the capital and resources to improve car sustainability—they just need to use it for the right things. These companies will continue to trailblaze, and hopefully in the right direction.
References and Sources
Gambarotta, A., Papetti, V., & Dimopoulos Eggenschwiler, P. (2019). Analysis of the effects of catalytic converter on automotive engines performance through real-time simulation models. Frontiers in Mechanical Engineering, 5. https://doi.org/10.3389/fmech.2019.00048
Laurenzi, M., & Spigler, R. (2018). Geometric effects in the design of catalytic converters in car exhaust pipes. Mathematics-in-Industry Case Studies, 9(1).https://doi.org/10.1186/s40929-018-0018-2
Toutov, A. A., Salata, M., Fedorov, A., Yang, Y.-F., Liang, Y., Cariou, R., Betz, K. N., Couzijn, E. P., Shabaker, J. W., Houk, K. N., & Grubbs, R. H. (2017). A potassium tert-butoxide and hydrosilane system for ultra-deep desulfurization of fuels. Nature Energy, 2(3). https://doi.org/10.1038/nenergy.2017.8