Promising Drug Against Pancreatic and Breast Cancers
Written by: Lily Song
Pancreatic cancer is caused by a type of cancerous growth that begins in the tissues of one’s pancreas — an organ in your abdomen that lies behind the lower part of your stomach. Breast cancer occurs when cells in the breast grow out of control. Twenty years ago, being diagnosed with pancreatic cancer was life-threatening news. Because of the discrete location of the pancreas behind the stomach, pancreatic cancer often cannot be detected until later stages. However, nowadays, with stronger support and funding, scientists have made large strides in treating pancreatic and breast cancer. Furthermore, the rate of breast cancer deaths in the U.S. has dramatically declined since 1950. As of 2017, the death rate from cancer had dropped from 31.9 to 19.9 per 100,000 population.
Recently, biology professors Zhi-Ren Liu and his team at Georgia State University developed a drug called ProAgio. They found that the drug was effective at treating pancreatic cancer and prolonging survival in mice, according to a study published in the journal Cellular and Molecular Gastroenterology and Hepatology. A second study, published in the Journal of Experimental Medicine, shows the drug is also effective against triple-negative breast cancer, which is a fast-growing and difficult-to-treat type of breast cancer. This type of breast cancer usually carries a poor prognosis because of its rapid growth.
Stroma
ProAgio is created from a human protein and targets the cell surface receptor integrin ?V??, which is expressed on cancer-associated fibroblasts. Fibroblasts are cells that generate collagen and other fibrous molecules. In tumors, it can create a thick barrier known as the stroma, which protects the cancer and helps it grow. The dense fibrotic stroma is what makes pancreatic cancer, which has a five-year survival rate of just eight percent, so lethal and difficult to treat. Among triple-negative breast cancer patients, research shows denser stroma is associated with poorer survival and high recurrence rates. Liu said that, “All solid tumors use cancer-associated fibroblasts, but in pancreatic cancer and triple-negative breast cancer, the stroma is so dense there’s often no way for conventional drugs to penetrate it and effectively treat the cancer”. The stroma also helps the tumor hide from one’s immune system. Immunotherapy, a type of treatment that uses your immune system to fight cancer, is less effective against tumors protected by a dense stroma filled with cancer-associated fibroblasts.
Figure 1
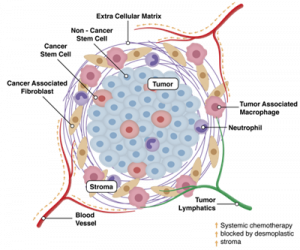
Depiction of a cancerous tumor (stroma identified)
Source: advancedchemotech.com
How It Works
ProAgio works by inducing apoptosis, or programmed cell death, in cancer-associated fibroblasts that express integrin ?V??. Cancer-associated fibroblasts promote angiogenesis, or the development of new blood vessels. Angiogenesis is crucial in aiding the spread of cancer because solid tumors need a blood supply to grow. In both studies, Liu and his team show that ProAgio has a profound effect on the arrangement of blood vessels in tumors. In pancreatic cancer, it reopened blood vessels that had collapsed due to high extravascular stress caused by the dense stroma. In triple-negative breast cancer, the drug’s anti-angiogenic activity reduced irregular angiogenic tumor vessels. In both cases, ProAgio allowed drugs to effectively reach the cancer.
ProAgio is unique because it only targets cancer-associated fibroblasts — a subclass of the cells that actively engage in supporting cancer — rather than inactive fibroblasts. This reduces side effects of the drug and increases its effectiveness. “When you have a wound, for example, normal fibroblasts will secrete fibers to limit the damage and promote healing,” said Liu. “The tumor region is basically a wound that won’t heal. Quiescent fibroblasts may play a role in preventing cancer from spreading. You don’t want to kill the good guys, only the bad guys.”
Next Steps
ProAgio is licensed to ProDa BioTech, a pharmaceutical research company founded by Liu. In 2018, ProDa BioTech received $2 million from the National Cancer Institute to fund toxicology and pharmacokinetic studies that are required before moving the drug to early-stage clinical trials. Those studies have been completed and the company has now submitted an Investigational New Drug (IND) Application, which is a request for authorization from the Food and Drug Administration to administer ProAgio to human subjects.
Once the IND is granted, the immediate next step is to begin clinical trials. The first trial will begin in early 2021 at the National Institute of Health Clinical Center in Bethesda, Md., and will be led by Christine Alewine, M.D., an oncologist at the National Cancer Institute. It’s purpose would be to determine patient tolerability and recommended phase II dose. In late 2021, Emory University is set to begin a multi-site trial among breast cancer and pancreatic cancer patients.
Conclusion
In conclusion, ProAgio has proven to be a promising drug for pancreatic and breast cancers. With more funding on these projects, great progress has been made in the treatment of these cancers – ProAgio being one of them. The thick stroma of a cancerous cell makes it especially difficult to target tumors. But, thankfully, ProAgio is unique in that it only targets cancer-associated fibroblasts instead of targeting the “bad” fibroblasts and the “good” fibroblasts, which would hinder tumor recession. This means that although it is difficult to “kill” cancer cells, Liu’s drug has hopes to be more efficient compared to other drugs. Finally, encouraging news on near-future clinical trials for the drug brighten the horizon for ProAgio.
References and Sources
Pancreatic cancer. (2020, April 24). Retrieved February 24, 2021, from https://www.mayoclinic.org/diseases-conditions/pancreatic-cancer/symptoms-causes/syc-20355421
Georgia State University. (2021, February 9). Drug is promising against pancreatic and breast cancers. ScienceDaily. Retrieved February 23, 2021 from www.sciencedaily.com/releases/2021/02/210209151856.htm
Weinstock, C. (2020, December 04). Advances in pancreatic cancer diagnosis and treatment. Retrieved February 24, 2021, from https://www.aarp.org/health/conditions-treatments/info-2020/advances-in-pancreatic-cancer-treatment.html